Qing Dai (QD), commonly known as Indigo naturalis, is a traditional Chinese medicine extracted from leaves and stems of plants like Indigofera tinctoria, Strobilanbthes cusia, O Kuntze, and Polygnonum tinctoium Lour. First mentioned in a foundational 10th-century Chinese medicine textbook, Qing Dai has been used for centuries.
Current research examines how components of Qing Dai interact with biological pathways and intestinal environments, contributing to ongoing academic interest in its molecular activity.
Mechanistic Research Focus
Current academic interest in Qing Dai centers on how plant-based compounds influence cytokines signaling, oxidative pathways, and receptor-mediated mechanisms. Research groups have explored how components of Qing Dai may:
- Influence production of cytokines such as TNF-α and IL-6.
-
Interact with NF-κB pathways
-
Affect oxidative-stress-related enzymes , including MPO
-
Act as ligands for the aryl hydrocarbon receptor (AhR)
-
Contribute to structural support of mucosal surfaces in experimental models
These mechanistic observations reflect laboratory and clinical-research curiosity, not therapeutic conclusions.
There are reported experiences—both anecdotal and in published studies—describing noticeable short-term changes among individuals using Qing Dai in supervised settings.
Because potency varies, QD should only be used under expert guidance and with appropriate follow-up.
The many anti-inflammatory mechanisms of QD
Qing Dai in Digestive-Health–Focused Research
A retrospective observational study examined the use of Qing Dai (QD) among individuals with digestive-tract–related issues who had been receiving care at Kyushu University Hospital. Clinical response and activity levels were assessed using the Rachmilewitz index or the internal activity index. The study included 17 individuals with large-intestine imbalance presentations and 8 individuals with small-intestine imbalance presentations.
At 8 weeks, reported observations included:
-
94.1% showing measurable changes (large-intestine group)
-
88.2% reaching the study’s predefined improvement category (large-intestine group)
-
37.5% showing measurable changes (small-intestine group)
Researchers described the patterns as “favorable” in the large-intestine group and “modest” in the small-intestine group.
QD to Support Individuals Exploring Natural Approaches
Clinical Efficacy and Safety of Oral Qing-Dai in Individuals in a Single-Center Open-Label Prospective Study
Researchers at Keio University Hospital evaluated oral Qing Dai among individuals experiencing moderate digestive difficulty.
20 participants with moderate to mild digestive activity were given Indigo naturalis twice a day (2g daily) for 8 weeks.
At week 8:
-
72% reported a clinical response
-
33% reached a clinician-defined improvement category
-
61% showed surface-level improvement
Clinical and internal scoring, CRP, and fecal occult blood results showed notable changes.
Researchers concluded Indigo naturalis was associated with meaningful shifts in these measurements.
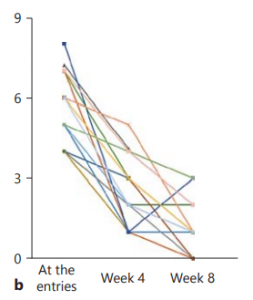
Efficacy of Indigo Naturalis in a Multicenter Randomized Controlled Trial
In 2016, researchers in Japan evaluated Indigo naturalis in 86 individuals with heightened digestive activity.
Groups were given 0.5g, 1.0g, or 2.0g daily for 8 weeks.
-
69.6% response in the 0.5 g group
-
75% in the 1.0 g group
-
81% in the 2.0 g group
After 8 weeks, participants receiving Qing Dai displayed higher rates of clinician-defined improvement than placebo.
60% of the 1.0 g group showed surface-level improvement.
Efficacy and Safety of Short-Term Therapy With Indigo Naturalis
This trial assessed short-term use of Qing Dai (QD) in individuals with mild to moderate digestive activity.
46 participants received either placebo or QD for 2 weeks.
Findings:
-
Placebo group showed no change
-
QD group showed improved Lichtiger index scoring and albumin levels
Researchers concluded that short-term QD produced measurable changes
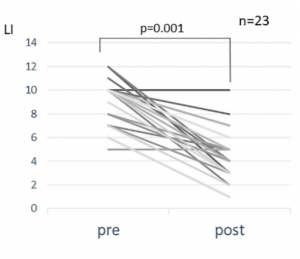
Evaluation of Qing Dai (QD)
This study observed 9 individuals experiencing heightened digestive difficulty who added Qing Dai alongside their existing routines.
Measurements:
-
Severity was evaluated with the Clinical Activity Index (CAI)
-
5 participants underwent internal imaging scoring
After 4 months:
-
CAI levels decreased
-
Some individuals using corticosteroids were able to discontinue them
Researchers reported significant shifts across multiple evaluation tools.
Development of an Indigo Naturalis (QD) Suppository
A prospective pilot study examining QD suppositories in 10 individuals with digestive irritation localized from the rectum to sigmoid.
Results at 4 weeks:
-
30% reached a clinician-defined improvement category
-
40% showed surface-level improvement
-
Rectal bleeding subscores improved
Efficacy of Indigo Naturalis (QD) Therapy for Intestinal Problems: A Case Series
This retrospective study reviewed 14 individuals taking QD.
After 8 weeks:
-
Partial Mayo scores decreased from 4 (2–5) to 1.5 (0–4)
-
50% showed clinical response
-
40% reached a clinician-defined improvement category
Qing-Dai for pediatric gut discomfort multicenter survey and systematic review
This review examined Qing Dai among 107 younger individuals.
Within 6 months:
-
80.2% reached a clinician-defined improvement category
Researchers noted significant observable changes.
Indigo Naturalis (QD) Effective for Treatment of Gut Issues With Steroid Dependency
This subanalysis evaluated QD vs. placebo for 8 weeks.
Observed responses:
-
77.8% in steroid-dependent participants
-
77.5% in those with prior anti–TNF-α use
-
70.8% with thiopurine
-
76.7% with high baseline imaging scores
QD groups showed higher rates of surface-level improvement and reduced fecal calprotectin.
Indigo Naturalis for Gut-Related Imbalance & Immune-Related Challenges
Open-label study of 20 individuals given 2g daily for 8 weeks.
At week 8:
-
72% showed clinical response
-
33% reached a clinician-defined improvement category
-
61% showed surface-level improvement
